Products
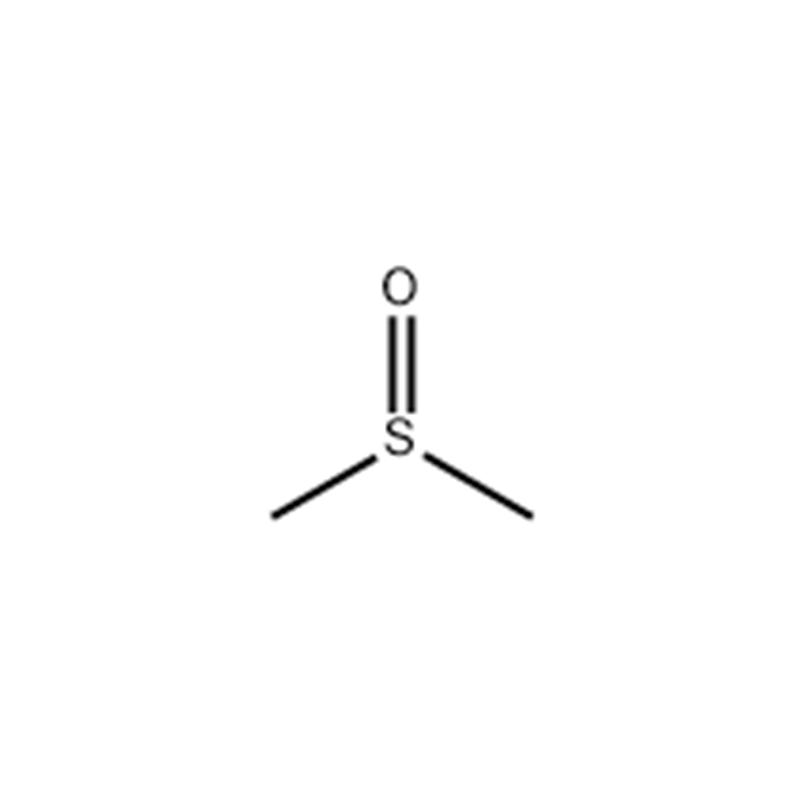
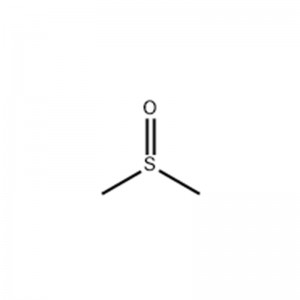
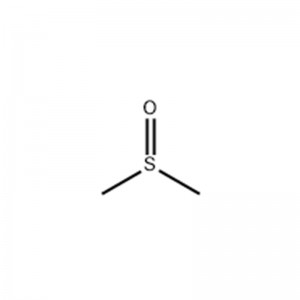
Methyl Sulfoxide
Structural Formula
Physical Properties
Appearance: colorless and odorless transparent liquid
Melting point:18.4°C
Boiling point:189°C(lit.)
Density:1.100g/mLat20°C
Vapor density:2.7(vsair)
Refractivity:n20/D1.479(lit.)
Flash point:192°F
Acidity coefficient(pKa):35(at25°C)
Relative polarity:0.444
Freezing point:18.4°C
Safety Data
It belongs to common goods
Customs code:2930300090
Export Tax Refund Rate(%):13%
Application
Dimethyl sulfoxide is widely used as solvent, reaction reagent and organic synthesis intermediate, especially as processing solvent and filament drawing solvent in acrylonitrile polymerization reaction, as solvent for polyurethane synthesis and filament drawing, as solvent for polyamide, polyimide and polysulfone resin, as well as solvent for aromatic hydrocarbon and butadiene extraction and solvent for synthesizing chlorofluoroaniline. Besides, dimethyl sulfoxide is directly used as the raw material and carrier of some drugs in the pharmaceutical industry. Dimethyl sulfoxide (DMSO) itself has anti-inflammatory, analgesic, diuretic and sedative effects, and is also known as a "panacea", which is often added to drugs as an active component of analgesic drugs.
Dimethyl sulfoxide (DMSO) is a sulfur-containing organic compound with the molecular formula C2H6OS, a colorless and odorless transparent liquid at room temperature, and a hygroscopic flammable liquid. It has the characteristics of high polarity, high boiling point, good thermal stability, non-protonic, miscible with water, soluble in most organic substances such as ethanol, propanol, benzene and chloroform, etc. It is known as "universal solvent". When heated in the presence of acid, a small amount of methyl mercaptan, formaldehyde, dimethyl sulfide, methanesulfonic acid and other compounds will be produced. It decomposes at high temperature, reacts violently with chlorine, and burns in air with light blue flame. It can be used as organic solvent, reaction medium and organic synthesis intermediate. It can also be used as dyeing solvent of synthetic fiber, dyeing agent, dyeing carrier and absorbent of recovering acetylene and sulfur dioxide.
Physical properties.
Colorless viscous liquid. Combustible, almost odorless, with bitter taste, hygroscopic. Except for petroleum ether, it can dissolve general organic solvents. Can be soluble in water, ethanol, acetone, acetaldehyde, pyridine, ethyl acetate, dibutyl benzodicarboxylate, dioxane and aromatic compounds, but insoluble in aliphatic hydrocarbon compounds other than acetylene. It has strong hygroscopicity and can absorb moisture equivalent to 70% of its own weight from the air at 20℃ when the relative humidity is 60%. The product is a weak oxidizer, and dimethyl sulfoxide without water is not corrosive to metals. When containing water, it is corrosive to iron; copper and other metals, but not to aluminum. Stable to bases. Heating in the presence of acid will produce a small amount of methyl mercaptan, formaldehyde, dimethyl sulfide; methanesulfonic acid and other compounds. Decomposition at high temperatures, can react violently with chlorine, burning in the air with a light blue flame.